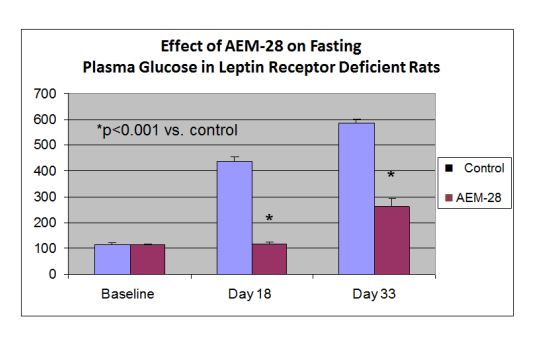
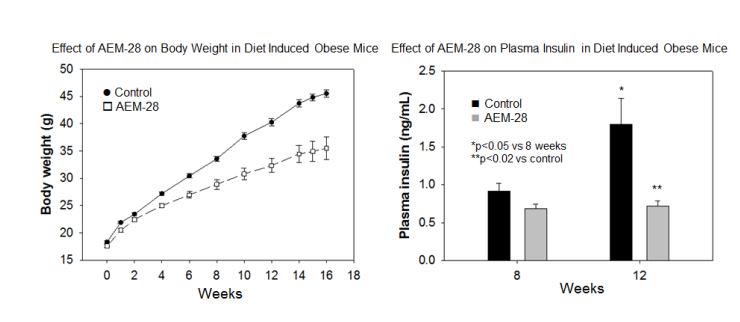
The newly allowed patent expands the utility of the CHAMP technology beyond claims in previous JV patents for the treatment of diabetic dyslipidemia and vascular diseases.
Capstone Therapeutics, Inc. (OTCQB:CAPS) (“the Company”) and its (60% owned) joint venture affiliate, LipimetiX Development, Inc. (“JV”), announced today the allowance of U.S. Patent application number 12/675,089 for the use of apolipoprotein E mimetic peptides to decrease plasma glucose in patients with diabetes or insulin resistance. In multiple animal models AEM-28, the parent peptide in the JV’s Chimeric Apolipoprotein E Mimetic Peptide (CHAMP) platform, increases insulin and glucose sensitivity, decreases plasma insulin and glucose and decreases weight gain in diet induced models of type 2 diabetes. The newly allowed patent expands the utility of the CHAMP technology beyond claims in previous JV patents for the treatment of diabetic dyslipidemia and vascular diseases.
“Macrophage infiltration of white adipose tissue in obesity contributes to a chronic inflammatory state resulting in altered cytokine secretion by the fat cells. The cytokine-created cross-talk between the inflamed adipose tissue, pancreas and liver results in altered metabolism, contributing to insulin resistance, elevated glucose and type 2 diabetes,” according to Dennis Goldberg, Ph.D., President of LipimetiX. “Our data indicate that the CHAMPs help normalize lipid metabolism, modulate the macrophage phenotype and decrease inflammation in the adipose tissue. The decrease in weight gain in the diet induced obese mice occurred despite comparable caloric intake during the study. These biologic activities result in statistically significant beneficial effects on plasma glucose and plasma insulin in validated preclinical models.”
“Increased formation of large, triglyceride rich VLDL particles and impaired clearance of these particles are hallmarks of insulin resistance and type 2 diabetes. AEM-28 decreased triglycerides and VLDL cholesterol by more than 70% within hours of single infusion in our recently reported Phase 1a and 1b/2a clinical studies,” said Goldberg. “The animal data supporting the new patent and the rapid clearance of VLDL cholesterol and triglycerides in human studies suggest that AEM-28 and other CHAMPs may provide a novel treatment modality for these patients.”
Charts accompanying this release are available at
http://media.globenewswire.com/cache/7986/file/38021.pdf
http://media.globenewswire.com/cache/7986/file/38022.pdf
Chimeric Apolipoprotein E Mimetic Peptides (CHAMPs)
Apolipoprotein E (Apo E) is in a class of protein that occurs throughout the body. Apo E is essential for the normal metabolism of cholesterol and triglycerides. After a meal, the postprandial (or post-meal) lipid load is packaged in lipoproteins and secreted into the blood stream. Apo E targets cholesterol and triglyceride rich lipoproteins to specific receptors in the liver, decreasing the levels in the blood. Elevated plasma cholesterol and triglycerides are independent risk factors for atherosclerosis, the buildup of cholesterol rich lesions and plaques in the arteries. Atherosclerosis is the major cause of cardiovascular disease, peripheral artery disease and cerebral artery disease, and can cause heart attack, loss of limbs and stroke. Defective lipid metabolism also plays an important role in the development of adult onset diabetes mellitus (Type 2 diabetes), and diabetics are particularly vulnerable to atherosclerosis, heart and peripheral artery diseases.
Scientists at the University of Alabama at Birmingham patented the first chimeric Apo E mimetic peptide in 1999, reducing the 299 amino acid native Apo E into a 28 amino acid, dual domain peptide that can be delivered therapeutically. One domain inserts into a lipoprotein surface and the second domain binds to the Apo E receptors. In 2010, our JV’s founding scientist, Dr. Dennis Goldberg, obtained worldwide rights to patents for Apo E mimetic peptides from the University of Alabama at Birmingham Research Foundation (“UABRF”). The JV has an Exclusive License Agreement with the UABRF for AEM-28, its analogs (CHAMPs), and uses.
About Capstone Therapeutics
Capstone Therapeutics is a biotechnology company committed to developing novel therapeutic peptides aimed at helping patients with under-served medical conditions. The Company is focused on development and commercialization of Chimeric Apo E Mimetic Peptides through the LipimetiX Development, Inc., joint venture.
Capstone’s corporate headquarters are in Tempe, Arizona. For more information, please visit the Company’s website: www.capstonethx.com. For more information on LipimetiX Development, please visit the JV’s website: www.lipimetix.com.
Statements in this press release or otherwise attributable to Capstone regarding our business that are not historical facts are made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. These forward-looking statements involve risks and uncertainties that could cause actual results to differ materially from predicted results. These risks include the factors discussed in our Form 10-K for the fiscal year ended December 31, 2014, and other documents we file with the U.S. Securities and Exchange Commission.
Editor’s Note: This press release is also available under the Investors section of the Company’s website at www.capstonethx.com.
FOR FURTHER INFORMATION: Investor Relations (602) 286-5250