- Roche collaborates with Merck to develop companion diagnostic for use with Keytruda (pembrolizumab), Merck’s anti-PD-1 therapy, in advanced solid tumors with mismatch repair deficiency (dMMR)
- Roche will seek FDA approval for biomarker panel expected to provide a standardized predictive testing option for dMMR
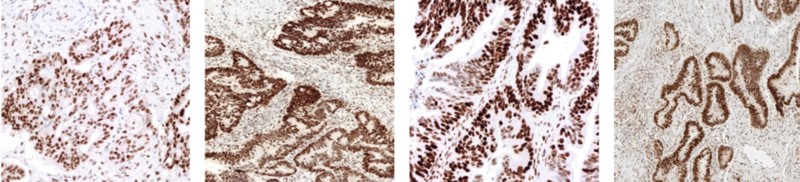
Colorectal cancer staining positive using the VENTANA MMR Panel (Image Courtesy of Roche Tissue Diagnostics)
Tucson, AZ, December 11, 2018
Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced that it entered into a collaboration with Merck (known as MSD outside the United States and Canada) to develop a companion diagnostic test to identify patients eligible for anti-PD-1 therapy based on the status of a biomarker in advanced solid tumors. The companies will collaborate on the development of a pan-cancer companion diagnostic to detect mismatch repair deficiency (dMMR) in solid tumors.
“We are excited to collaborate with Merck to develop a pan-cancer companion diagnostic test panel to detect mismatch repair deficiency,” said Jill German, Head of Roche Tissue Diagnostics. “This new development could help change the way we identify patients best suited for immunotherapy treatment.”
Cancer treatment has been predominantly determined by the location in the body where the tumor originated. The development of an MMR immunohistochemistry (IHC) assay potentially paves the way for treatment decisions to be made based on biomarker expression within solid tumors originating from various parts of the body.
“A key element of our strategy at Merck is focused on identifying those patients likely to benefit most from our medicines,” said Dr. Eric Rubin, senior vice president, oncology clinical development, Merck Research Laboratories. “We look forward to working with Roche to develop a diagnostic test for mismatch repair deficiency.”
In May 2017, Keytruda®, Merck’s anti-PD-1 therapy, became the first cancer treatment approved by the US Food and Drug Administration for the treatment of adult and pediatric patients with unresectable or metastatic, microsatellite instability-high or mismatch repair deficient solid tumors that have progressed following prior treatment and who have no other alternative treatment options, or colorectal cancer that has progressed following treatment with certain chemotherapy drugs.
Testing for MMR consists of the immunohistochemical detection of four MMR proteins (MLH1, MSH2, MSH6 and PMS2).1,2 These assays, along with BRAF V600E, are currently used clinically to aid in the identification of a genetic predisposition for colorectal and other cancers called Lynch syndrome. This collaboration is expected to expand the utility of this panel to include selection of patients with solid tumors for immunotherapy.
The companion diagnostic currently under development is an IHC test for use on the Roche BenchMark ULTRA instrument, which, as the most widely installed IHC/ISH (in situ hybridization) staining platform globally, will provide broad testing access to patients.

About Roche
Roche is a global pioneer in pharmaceuticals and diagnostics focused on advancing science to improve people’s lives. The combined strengths of pharmaceuticals and diagnostics under one roof have made Roche the leader in personalised healthcare – a strategy that aims to fit the right treatment to each patient in the best way possible.
Roche is the world’s largest biotech company, with truly differentiated medicines in oncology, immunology, infectious diseases, ophthalmology and diseases of the central nervous system. Roche is also the world leader in in vitro diagnostics and tissue-based cancer diagnostics, and a frontrunner in diabetes management. Founded in 1896, Roche continues to search for better ways to prevent, diagnose and treat diseases and make a sustainable contribution to society. The company also aims to improve patient access to medical innovations by working with all relevant stakeholders. Thirty medicines developed by Roche are included in the World Health Organization Model Lists of Essential Medicines, among them life-saving antibiotics, antimalarials and cancer medicines. Moreover, for the tenth consecutive year, Roche has been recognised as the most sustainable company in the Pharmaceuticals Industry by the Dow Jones Sustainability Indices (DJSI).
The Roche Group, headquartered in Basel, Switzerland, is active in over 100 countries and in 2017 employed about 94,000 people worldwide. In 2017, Roche invested CHF 10.4 billion in R&D and posted sales of CHF 53.3 billion. Genentech, in the United States, is a wholly owned member of the Roche Group. Roche is the majority shareholder in Chugai Pharmaceutical, Japan. For more information, please visit www.roche.com.
VENTANA and BENCHMARK are trademarks of Roche. KEYTRUDA is a registered trademark of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA. Other product names and trademarks are the property of their respective owners.
References
[1] Bartley AN, Hamilton SR, Alsabeh R, et al. Template for reporting results of biomarker testing of specimens from patients with carcinoma of the colon and rectum. Arch Pathol Lab Med. 2014;138(2):166–170.
[2] Vilar E, Gruber SB. Microsatellite instability in colorectal cancer—the stable evidence. Nat Rev Clin Oncol. 2010;7(3):153–162.
Colorectal cancer staining positive using the VENTANA MMR Panel
Roche Tissue Diagnostics Media Relations
Gabrielle Fimbres
Phone: 520.222.4573
gabrielle.fimbres@roche.com
© 2018 Roche Tissue Diagnostics
All trademarks used or mentioned in this release are protected by law.
