Having good eyesight is a gift. When age or health issues affect our vision, life science innovations as prescribed by our healthcare professionals help us improve our vision and protect the health of our eyes.
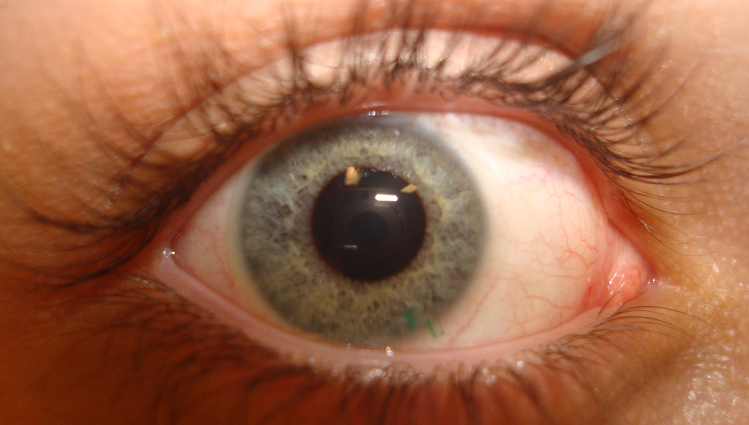
Contact lenses, other than the cosmetic variety, become almost invisible once inserted in the eye. Most corrective contact lenses come with a light “handling tint” that may render the lens slightly more visible on the eye. Soft contact lenses extend beyond the cornea, and the border is sometimes visible against the sclera. – Source: Wikipedia
A History of Innovation: Contact Lenses
The concept of placing a corrective lens on the eye dates all the way back to 1508 when Leonardo da Vinci illustrated the concept of contact lenses. Three hundred years later British astronomer Sir John Herschel conceptualizes practical lens design in 1823 and in 1939 the first plastic contact lenses came to market. Innovation continued with the the introduction of soft contact lenses in 1971 and FDA approval of new soft contact lenses for extended (overnight) wear in 1981. (Source: http://www.contactlenses.org/timeline.htm)
Today, innovation continues as life science innovators continue to work to make new products and devices that make contact lenses better for patients globally.
 Your Eye Health Partnership: You and Your Eye Care Professional
Your Eye Health Partnership: You and Your Eye Care Professional
Unlike eyeglasses that are usually about 12 millimeters from the eye, the contact lens is positioned directly on the tear film of the eye.
Here in Arizona, contact lens wearers visit their ophthalmologist or optometrist to have a complete eye exam every year and get an updated prescription.
This is important as your eye care professional will not only make sure that your contact lens are fitting correctly and that your prescription has not changed, but more importantly, they will check your eyes for serious conditions such as glaucoma, diabetes, uveitis, hypertension, macular degeneration, and retina problems just to name a few. Also, even a small change in your prescription can lead to headaches and eye strain if not corrected even if you feel that you can see just fine with your current prescription. So, in summary, the health of your eyes is not something to scrimp on. Discuss your financial concerns with your provider as there are often ways to help if money is tight. Also your eye care professional will be able to explain why it is important for you to not go longer than a year without a complete eye health check up and what you need to do between visits so that you get the best possible results from contact lens wear.
Adherence: Sticking with your eye care professional’s instructions
Adherence in the case of healthcare innovations like prescription drugs, medical devices, and contact lenses, means to faithfully follow your health care professional’s instructions. When used as directed and properly monitored regularly on the schedule recommended by healthcare professionals, life science innovations produce quality outcomes and make our life better.
Quality healthcare outcomes depend upon patients’ adherence to recommended treatment regimens. Patient nonadherence can be a pervasive threat to health and wellbeing and carry an appreciable economic burden as well. In some disease conditions, more than 40% of patients sustain significant risks by misunderstanding, forgetting, or ignoring healthcare advice. While no single intervention strategy can improve the adherence of all patients, decades of research studies agree that successful attempts to improve patient adherence depend upon a set of key factors. These include realistic assessment of patients’ knowledge and understanding of the regimen, clear and effective communication between health professionals and their patients, and the nurturance of trust in the therapeutic relationship. (Source: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1661624/)
The CDC in 2014 compiled a report showing that there are at least 1 million physician or emergency room visits every year in the US for contact lens issues. These medical conditions include: keratitis, corneal ulcers, corneal neovascularization, papillary conjunctivitis, and corneal scarring resulting in permanent vision loss and others. Many of these issues can be avoided with proper care of the lenses by the patient. Nearly 1 million US health care visits for keratitis occur annually at a cost of $175 million. One third of contact lens wearers report at least one health care visit for a red or painful eye while wearing contact lenses.
Contact Lenses and the Law:
This year at the Arizona Legislature, a bill proposes to extend the length of a contact lens prescription beyond one year. This is not a good idea when it comes to ensuring that patients get the best possible outcome from their contact lenses. This proposed change is contrary to the vast majority of contact lens prescription laws in the United States, and is contrary to the underlying prescriptions laws for all prescription pharmaceuticals in Arizona.
Arizona statutes provide that a contact lens prescription cannot exceed one year. This is consistent with the laws of at least 42 states. In states where prescriptions are allowed for a longer period of time, the Arizona Optometric Association has verified that the actual standard for contact lens prescriptions in those states is one year. In consulting with lead pharmacists in Arizona, the one year prescription for all pharmaceuticals and for contact lenses is based on medical practice, i.e., that a patient should be reevaluated on a regular periodic basis for the condition for which the pharmaceutical (or medical device) was prescribed, and whether the prescription or medical device is effective and is not causing other conditions.
There is both federal and state law on the subject of contact lenses. Federal law provides a mechanism for the sale of contact lenses by internet. However, federal law specifically refers to and incorporates state contact lens prescription requirements which are valid if they are one year or longer, except for doctor-prescribed exceptions. Therefore, under federal law, a one year prescription limit is fully acceptable. The U.S. Center for Disease Control, the American Academy of Ophthalmology, the American Optometric Association and contact lens manufacturers all recommend annual patient exams for patients who wear contact lenses.
Some hard and some soft contact lenses are a Class III medical device because they are extended use; single use contact dailies and other soft or hard lenses that are removed daily are a Class II medical device. Class II medical devices require regulation to provide a reasonable assurance and safety and effectiveness (Class II devices include fusion pumps, x-ray systems, etc.). Most contact lens wearers occasionally sleep in the Class II lenses as determined by a CDC study last year. Extended wear contact lenses are Class III FDA medical devices. They are in this category because they pose a higher risk of injury or illness and are subject to free market approval and increased regulation. (Examples of other Class III medical devices include heart valves, pacemakers, dental lasers and intraocular lenses.)
