Interim analysis of original cohorts of Phase 1 study evaluated two-dose vaccination schedule of mRNA-1273 across three dose levels (25, 100, 250 µg) in 45 healthy adults ages 18-55 years; results reaffirm and expand upon positive interim data announced on May 18th
- Neutralizing antibody titers were observed in 100% of evaluated participants; at the 100 µg dose level selected for Phase 3, the geometric mean titers were above those seen in convalescent sera
- Vaccination with mRNA-1273 elicited Th1-biased CD4 T cell responses
- mRNA-1273 was generally safe and well-tolerated
- Data support 30,000 participant Phase 3 study expected to begin on July 27
- Conference call to be held on Wednesday, July 15 at 8:00 a.m. ET
 CAMBRIDGE, Mass.–(BUSINESS WIRE)–Jul. 14, 2020– Moderna, Inc., (Nasdaq:MRNA) a clinical stage biotechnology company pioneering messenger RNA (mRNA) therapeutics and vaccines to create a new generation of transformative medicines for patients, today announced the publication of an interim analysis of the open-label Phase 1 study of mRNA-1273, its vaccine candidate against COVID-19, in The New England Journal of Medicine. This interim analysis evaluated a two-dose vaccination schedule of mRNA-1273 given 28 days apart across three dose levels (25, 100, 250 µg) in 45 healthy adult participants ages 18-55 years, and reports results through Day 57. Results from participants in the initial dose cohorts who received both vaccinations and were evaluated at pre-specified timepoints reaffirm the positive interim data assessment announced on May 18th and show mRNA-1273 induced rapid and strong immune responses against SARS-CoV-2. The study was led by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health (NIH).
CAMBRIDGE, Mass.–(BUSINESS WIRE)–Jul. 14, 2020– Moderna, Inc., (Nasdaq:MRNA) a clinical stage biotechnology company pioneering messenger RNA (mRNA) therapeutics and vaccines to create a new generation of transformative medicines for patients, today announced the publication of an interim analysis of the open-label Phase 1 study of mRNA-1273, its vaccine candidate against COVID-19, in The New England Journal of Medicine. This interim analysis evaluated a two-dose vaccination schedule of mRNA-1273 given 28 days apart across three dose levels (25, 100, 250 µg) in 45 healthy adult participants ages 18-55 years, and reports results through Day 57. Results from participants in the initial dose cohorts who received both vaccinations and were evaluated at pre-specified timepoints reaffirm the positive interim data assessment announced on May 18th and show mRNA-1273 induced rapid and strong immune responses against SARS-CoV-2. The study was led by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health (NIH).
mRNA-1273 was generally safe and well-tolerated, with no serious adverse events reported through Day 57. Adverse events (AEs) were generally transient and mild to moderate in severity. The most notable adverse events were seen at the 250 µg dose level, with three of those 14 participants (21%) reporting one or more severe events. Solicited systemic adverse events were more common after the second vaccination and occurred in seven of 13 (54%) participants in the 25 µg group, all 15 participants in the 100 µg group and all 14 participants in the 250 µg group. The most commonly reported systemic adverse events following second vaccination at the 100 µg dose were fatigue (80%), chills (80%), headache (60%) and myalgia (53%), all of which were transient and mild or moderate in severity. The most common solicited local adverse event at the 100 µg dose was pain at the injection site (100%), which was also transient and mild or moderate in severity. Evaluation of clinical safety laboratory values grade 2 or higher and unsolicited adverse events revealed no patterns of concern.
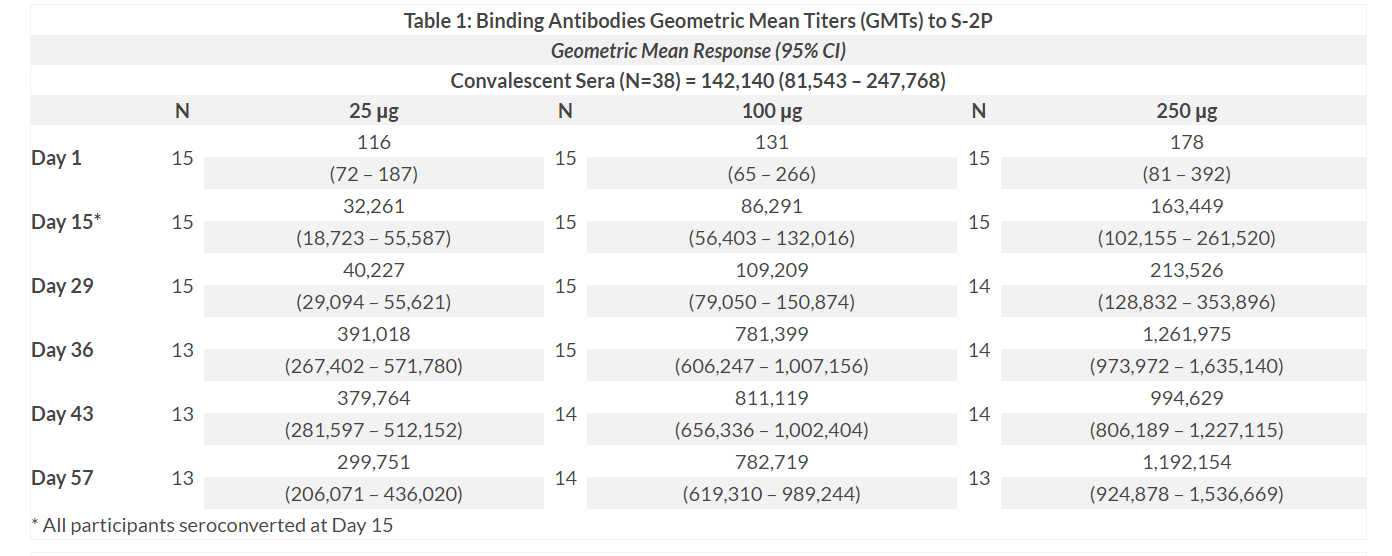
mRNA-1273 induced binding antibodies to the full-length SARS-CoV-2 Spike protein (S) in all participants after the first vaccination, with all participants seroconverting by Day 15. Dose dependent increases in binding titers were seen across the three dose levels, and between prime and boost vaccinations within the dose cohorts. After two vaccinations, at Day 57, geometric mean titers exceeded those seen in convalescent sera obtained from 38 individuals with confirmed COVID-19 diagnosis 1. Of the 38 individuals in the convalescent sera group, 15% were classified as having severe symptoms (hospitalization requiring intensive care and/or ventilation), 22% had moderate symptoms and 63% had mild symptoms. Convalescent sera samples were tested using the same assays as the study samples.
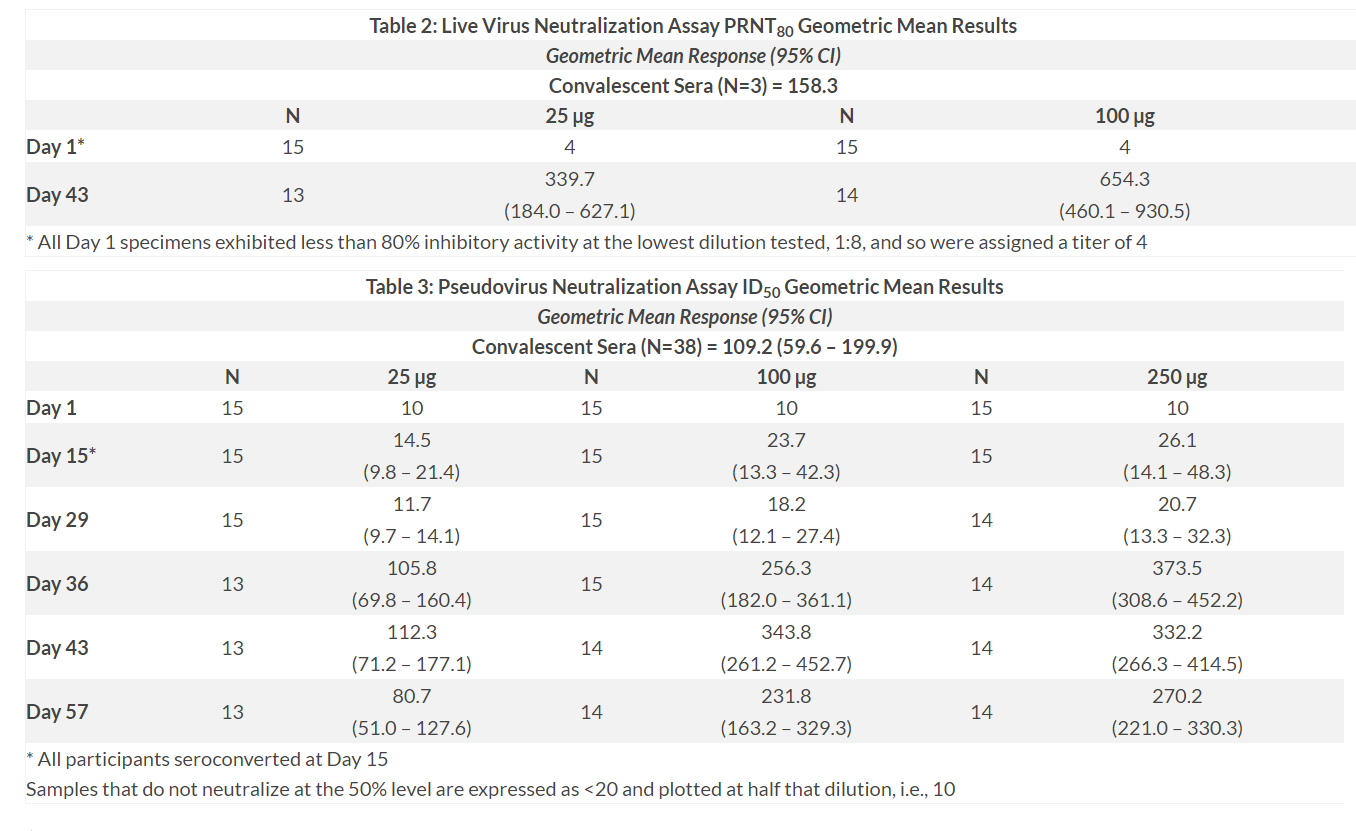
Neutralizing activity was assessed in two different assays, a live SARS-CoV-2 plaque-reduction neutralization test (PRNT) and a pseudovirus neutralization assay (pseudotyped lentivirus reporter single-round-of-infection neutralization assay, PsVNA). No participants had detectable live SARS-CoV-2 virus neutralization or PsVNA responses prior to vaccination.
After two vaccinations, mRNA-1273 elicited robust neutralizing antibody titers. At Day 43, neutralizing activity against SARS-CoV-2 (PRNT80) was seen in all evaluated participants2. At the Phase 3 selected dose of 100 µg, the geometric mean titer levels were 4.1-fold above those seen in reference convalescent sera (n=3).
After the second vaccination, PsVNA neutralizing antibody titers were detected in all participants in all dose cohorts. The Day 57 geometric mean titers at the 100 µg dose were 2.1-fold higher than those seen in convalescent sera (n=38)3. Strong correlations were observed between the binding and neutralization assays, and between the live virus and pseudovirus neutralization assays. A clear dose response was seen in geometric mean titers between the 25 µg and 100 µg dose levels, with minimal additional increases at the 250 µg dose.
T-cell responses were also evaluated at the 25 µg and 100 µg dose levels. Following second vaccination, mRNA-1273 elicited Th1-biased CD4 T-cell responses without significant elevation of Th2-biased CD4 T-cell responses.
Evaluation of the durability of immune responses is ongoing, and participants will be followed for one year after the second vaccination, with scheduled blood collections throughout that period. An additional seven cohorts in this Phase 1 study have completed enrollment: a 50 µg cohort in adults 18-55 (n=15), three cohorts of older adults (n=30, ages 56-70, 25 µg, 50 µg, and 100 µg) and three cohorts of elderly adults (n=30, ages 71 and above, 25 µg, 50 µg, and 100 µg). These data are expected to be published separately. Given the increased morbidity and mortality of COVID-19 in older and elderly adults, additional evidence for the potential of a vaccine to protect this population is an urgent priority.
“These Phase 1 data demonstrate that vaccination with mRNA-1273 elicits a robust immune response across all dose levels and clearly support the choice of 100 µg in a prime and boost regimen as the optimal dose for the Phase 3 study,” said Tal Zaks, M.D., Ph.D., Chief Medical Officer of Moderna. “We look forward to beginning our Phase 3 study of mRNA-1273 this month to demonstrate our vaccine’s ability to significantly reduce the risk of COVID-19 disease.”
“These positive Phase 1 data are encouraging and represent an important step forward in the clinical development of mRNA-1273, our vaccine candidate against COVID-19, and we thank the NIH for their ongoing collaboration. The Moderna team continues to focus on starting our Phase 3 study this month and, if successful, filing a BLA,” said Stéphane Bancel, Chief Executive Officer of Moderna. “We are committed to advancing the clinical development of mRNA-1273 as quickly and safely as possible while investing to scale up manufacturing so that we can help address this global health emergency.”
Both cohorts, healthy adults ages 18-55 years (n=300) and older adults ages 55 years and above (n=300), in the Company’s Phase 2 study of mRNA-1273 are fully enrolled. This Phase 2 placebo-controlled, dose-confirmation study is evaluating the safety, reactogenicity and immunogenicity of two vaccinations of mRNA-1273 given 28 days apart. Each participant is receiving placebo, a 50 μg or a 100 μg dose at both vaccinations.
The Phase 3 study protocol has been reviewed by the U.S. Food and Drug Administration (FDA) and is aligned to recent FDA guidance on clinical trial design for COVID-19 vaccine studies. The randomized, 1:1 placebo-controlled trial is expected to include approximately 30,000 participants at the 100 µg dose level in the U.S. The primary endpoint will be the prevention of symptomatic COVID-19 disease. Key secondary endpoints include prevention of severe COVID-19 disease (as defined by the need for hospitalization) and prevention of infection by SARS-CoV-2. The primary efficacy analysis will be an event-driven analysis based on the number of participants with symptomatic COVID-19 disease. The target vaccine efficacy (VE) against COVID-19 for powering assumptions is 60% (95% confidence interval to exclude a lower bound >30%). Data will be reviewed by an independent Data Safety Monitoring Board organized by NIH. The trial is expected to have two interim analyses (at approximately 53 and 106 events), prior to a final event-driven analysis, at approximately 151 events. The primary efficacy analysis will be an event-driven analysis based on the number of participants with symptomatic COVID-19 disease. This Phase 3 study has been named the COVE study. The ClinicalTrials.gov identifier is NCT04470427. Registration for the COVE study is expected to be available at study centers across the country beginning on July 21 with study initiation on July 27.
Moderna is working closely with Operation Warp Speed (OWS) and the NIH, including NIAID’s COVID-19 Prevention Trials Network (COVPN), to conduct the Phase 3 COVE study. Working together with collaborators like NIH, the Company hopes to achieve a shared goal that the participants in the COVE study are representative of the communities at highest risk for COVID-19 and of our diverse society.
Moderna has completed manufacture of vaccine required to start the Phase 3 study. With the Phase 3 dose being finalized at 100 μg, the Company remains on track to be able to deliver approximately 500 million doses per year, and possibly up to 1 billion doses per year, beginning in 2021 from the Company’s internal U.S. manufacturing site and strategic collaboration with Lonza. In addition, Moderna recently announced a collaboration with Catalent for large-scale, commercial fill-finish manufacturing of mRNA-1273 at Catalent’s biologics facility in Indiana. On July 9, Moderna announced a collaboration with ROVI for large-scale, commercial fill-finish manufacturing of mRNA-1273 intended in principle to supply markets outside of the U.S. starting in early 2021 at ROVI’s facility in Madrid, Spain.
Funding from the Biomedical Advanced Research and Development Authority (BARDA), a division of the Office of the Assistant Secretary for Preparedness and Response (ASPR) within the U.S. Department of Health and Human Services (HHS), partially supported the planning for the Phase 2 and Phase 3 studies of mRNA-1273 and is supporting the execution of these studies, as well as the manufacturing process scale-up of mRNA-1273. Moderna will also fund costs required to complete the development of mRNA-1273 including portions of the Phase 3 study and the scale up of manufacturing capacity at the final established dosage in order to obtain licensure for mRNA-1273. A summary of the company’s work to date on COVID-19 can be found here.
Data tables can be found at the bottom of this press release.
Conference Call and Webcast Information
Moderna will host a live conference call and webcast at 8:00 a.m. ET on Wednesday, July 15, 2020. To access the live conference call, please dial 866-922-5184 (domestic) or 409-937-8950 (international) and refer to conference ID 6345699. A webcast of the call will also be available under “Events and Presentations” in the Investors section of the Moderna website at investors.modernatx.com. The archived webcast will be available on Moderna’s website approximately two hours after the conference call.
______________
1 Refer to Table 1 below.
2 Refer to Table 2 below.
3 Refer to Table 3 below.
About mRNA-1273
mRNA-1273 is an mRNA vaccine against COVID-19 encoding for a prefusion stabilized form of the Spike (S) protein, which was selected by Moderna in collaboration with investigators from the VRC. The first clinical batch, which was funded by the Coalition for Epidemic Preparedness Innovations, was completed on February 7, 2020 and underwent analytical testing; it was shipped to NIH on February 24, 42 days from sequence selection. The first participant in the NIAID-led Phase 1 study of mRNA-1273 was dosed on March 16, 63 days from sequence selection to Phase 1 study dosing. On May 12, the FDA granted mRNA-1273 Fast Track designation. Both cohorts, healthy adults ages 18-55 years (n=300) and older adults ages 55 years and above (n=300), in the Company’s Phase 2 study of mRNA-1273 are fully enrolled.
About Moderna’s Prophylactic Vaccines Modality
Moderna scientists designed the company’s prophylactic vaccines modality to prevent infectious diseases. More than 1,900 participants have been enrolled in Moderna’s infectious disease vaccine clinical studies under health authorities in the U.S., Europe and Australia. Clinical data demonstrate that Moderna’s proprietary vaccine technology has been generally well-tolerated and can elicit durable immune responses to viral antigens. Based on clinical experience across Phase 1 studies, the company designated prophylactic vaccines a core modality and is working to accelerate the development of its vaccine pipeline.
The potential advantages of an mRNA approach to prophylactic vaccines include the ability to combine multiple mRNAs into a single vaccine, rapid discovery to respond to emerging pandemic threats and manufacturing agility derived from the platform nature of mRNA vaccine design and production. Moderna has built a fully integrated manufacturing plant which enables the promise of the technology platform.
Moderna currently has nine development candidates in its prophylactic vaccines modality, including:
- Vaccines against respiratory infections
- Respiratory syncytial virus (RSV) vaccine for older adults (mRNA-1777 and mRNA-1172 or V172 with Merck)
- RSV vaccine for young children (mRNA-1345)
- Human metapneumovirus (hMPV) and parainfluenza virus type 3 (PIV3) vaccine (mRNA-1653)
- COVID-19 vaccine (mRNA-1273)
- Influenza H7N9 (mRNA-1851)
- Vaccines against infections transmitted from mother to baby
- Cytomegalovirus (CMV) vaccine (mRNA-1647)
- Zika vaccine (mRNA-1893 with BARDA)
- Vaccines against highly prevalent viral infections
- Epstein-Barr virus (EBV) vaccine (mRNA-1189)
- To date, Moderna has demonstrated positive Phase 1 data readouts for eight prophylactic vaccines (H10N8, H7N9, RSV, chikungunya virus, hMPV/PIV3, CMV, Zika and COVID-19). Moderna’s CMV vaccine is currently in a Phase 2 dose-confirmation study. Moderna’s investigational Zika vaccine (mRNA-1893), currently in a Phase 1 study, was granted FDA Fast Track designation in August 2019.
About Moderna
Moderna is advancing messenger RNA (mRNA) science to create a new class of transformative medicines for patients. mRNA medicines are designed to direct the body’s cells to produce intracellular, membrane or secreted proteins that can have a therapeutic or preventive benefit and have the potential to address a broad spectrum of diseases. The company’s platform builds on continuous advances in basic and applied mRNA science, delivery technology and manufacturing, providing Moderna the capability to pursue in parallel a robust pipeline of new development candidates. Moderna is developing therapeutics and vaccines for infectious diseases, immuno-oncology, rare diseases and cardiovascular diseases, independently and with strategic collaborators.
Headquartered in Cambridge, Mass., Moderna currently has strategic alliances for development programs with AstraZeneca PLC and Merck & Co., Inc., as well as the Defense Advanced Research Projects Agency (DARPA), an agency of the U.S. Department of Defense, and the Biomedical Advanced Research and Development Authority (BARDA), a division of the Office of the Assistant Secretary for Preparedness and Response (ASPR) within the U.S. Department of Health and Human Services (HHS). Moderna has been ranked in the top ten of Science’s list of top biopharma industry employers for the past five years. To learn more, visit www.modernatx.com.
Forward Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including regarding the Company’s development of a potential vaccine against the novel coronavirus, the parameters of the Phase 1 and Phase 2 studies of mRNA-1273, the publication of study data for later cohorts in the Phase 1 study of mRNA-1273, the parameters and timing of the Phase 3 study of mRNA-1273, the potential filing of a biologics license application (BLA) for mRNA-1273, the Company’s potential manufacturing capabilities and projected vaccine dose production, and costs related to the mRNA-1273 program to be funded by the Company. In some cases, forward-looking statements can be identified by terminology such as “will,” “may,” “should,” “could”, “expects,” “intends,” “plans,” “aims,” “anticipates,” “believes,” “estimates,” “predicts,” “potential,” “continue,” or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words. The forward-looking statements in this press release are neither promises nor guarantees, and you should not place undue reliance on these forward-looking statements because they involve known and unknown risks, uncertainties, and other factors, many of which are beyond Moderna’s control and which could cause actual results to differ materially from those expressed or implied by these forward-looking statements. These risks, uncertainties, and other factors include, among others: the fact that there has never been a commercial product utilizing mRNA technology approved for use; the fact that the rapid response technology in use by Moderna is still being developed and implemented; the fact that the safety and efficacy of mRNA-1273 has not yet been established; potential adverse impacts due to the global COVID-19 pandemic such as delays in regulatory review, manufacturing and clinical trials, supply chain interruptions, adverse effects on healthcare systems and disruption of the global economy; and those other risks and uncertainties described under the heading “Risk Factors” in Moderna’s most recent Quarterly Report on Form 10-Q filed with the U.S. Securities and Exchange Commission (SEC) and in subsequent filings made by Moderna with the SEC, which are available on the SEC’s website at www.sec.gov. Except as required by law, Moderna disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this press release in the event of new information, future developments or otherwise. These forward-looking statements are based on Moderna’s current expectations and speak only as of the date hereof.
Data Tables
View source version on businesswire.com: https://www.businesswire.com/news/home/20200714006042/en/
Moderna
Media:
Colleen Hussey
Senior Manager, Corporate Communications
203-470-5620
Colleen.Hussey@modernatx.com
Dan Budwick
1AB
973-271-6085
Dan@1abmedia.com
Investors:
Lavina Talukdar
Head of Investor Relations
617-209-5834
Lavina.Talukdar@modernatx.com
Source: Moderna, Inc